Before COVID-19, figuring out whether a sore throat, fever, and runny nose were caused by a cold, flu, or strep wasn’t a top priority. You either powered through, knowing you’d be miserable for a few days but would probably feel better soon or you visited the doctor’s office, urgent care, or emergency room where you might get a test to figure out which virus or bacteria was behind your misery, and maybe a prescription to treat it. But even doctors often don’t order tests, preferring instead to make diagnoses based on symptoms.
During the pandemic, however, it became more critical to know who was infected with SARS-CoV-2 so those people could isolate and not spread the virus. At-home test kits became household products, and most people are by now adept at squeezing out a few drops of solution onto a card, sticking a swab up their nose, swirling the swab in the testing liquid, and waiting for a result.
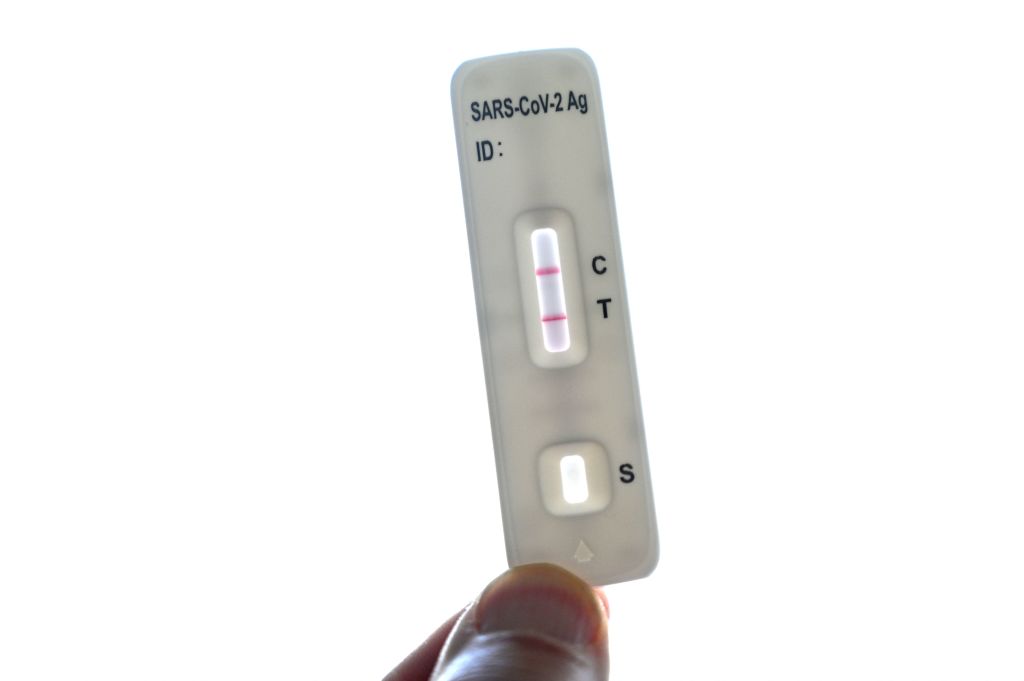
Tests for COVID-19
A number of companies, offer at-home test kits for COVID-19 that people can buy without a prescription. Most of these are rapid antigen tests, which pick up antigens, or substances that the SARS-CoV-2 virus makes. While they are relatively accurate, because it takes some time for levels of these antigens to build up after an infection, these tests are less reliable in the day or so immediately after exposure to the virus. That’s why doctors recommend repeating the test a couple of days apart.
Then there are PCR, or molecular, tests, that are closer to the gold standard used in laboratories, and rely on finding the smallest genetic signatures of the virus and amplifying them. Cue Health sells a palm-sized COVID-19 PCR device that people can purchase for $300 that allows for 10 at-home tests and doesn’t require a prescription. The device reads a saliva sample and provides results to a smartphone app in 20 minutes.
Other testing companies, such as Labcorp and Everlywell, also sell PCR-based tests for COVID-19, but they are technically collection kits, which means that you have to send a saliva sample into a lab to get results, which can mean critical days will pass before you get results.
Tests for seasonal flu
There is only one at-home test authorized by the FDA for influenza: Lucira, now owned by Pfizer, makes a combined flu and COVID-19 molecular test. The kit uses a PCR-related method of amplifying and reading any viral genes in a sample, providing a result in about 30 minutes. In coming months, eMed, a digital health company that sells test kits and telehealth services, plans to offer that kit, as well another that is currently being reviewed by the U.S. Food and Drug Administration. (The combined kit currently available on eMed’s website only tests for COVID-19 and provides a survey to help telemedicine professionals determine if a user likely has the flu.)
Tests for RSV
There currently aren’t at-home tests for RSV, because typically only infants and the elderly are at risk for severe illness. Doctors’ offices, hospitals, and urgent care centers can perform RSV tests that provide results in about 30 minutes.
The advantages of at-home testing
The currently cumbersome journey from symptoms to treatment rests on people making appointments with their doctors or at an urgent care center and could be contributing to the spread of viruses. Self-testing could reduce some of that risk since people are most infectious while they are waiting to get tested and receive treatment. Ideally, if someone tests themselves at home and finds they are positive for influenza or COVID-19, for example, they don’t necessarily have to physically visit their doctor or go into an urgent care or emergency room, where they could spread the virus to others. Instead, they could video conference with a doctor who could assess their symptoms and at-home test results, then write a prescription for, say, oseltamivir if it’s the flu, or Paxlovid if it’s COVID-19. “If you’re trying to minimize morbidity and mortality, and cut down on transmission, then [at-home] testing is an incredible tool to have,” says Thomas Denny, chief operating officer of the Duke University Human Vaccine Institute.
Read more: What It Will Take to Avoid a Tripledemic This Winter
In addition to the altruistic public health reasons to test yourself, there are more selfish ones. Knowing what infection you have means you can get the right treatment, and hopefully avoid prolonged and serious illness. And for the first time, there are effective ways to manage all three major respiratory illnesses that dominate the winter: flu, COVID-19, and respiratory syncytial virus (RSV). Self-testing kits are especially useful for influenza and COVID-19, since antiviral treatments for both are most effective when people taken them within the first few days after their symptoms appear. (And while there aren’t treatments for RSV, new vaccines and antibody treatments can help to prevent infections and potentially serious complications in the most vulnerable.) Waiting to make an appointment to get tested at a health facility means many people could miss this small treatment window. “A lot of people are happy with the status quo, and don’t see a need for self-testing,” says Dr. Michael Mina, chief science officer at eMed.
And the availability of COVID-19 kits has set new expectations for at-home testing, leading both the public and health professionals to ask why more people can’t test for other illnesses themselves. In many cases, “we don’t need people to come into doctors’ offices,” says Dr. Howard Heller, an infectious disease specialist at Massachusetts General Hospital and Harvard Medical School. “The home testing space is evolving and snowballing.”
During the pandemic, the Biden Administration launched the Test to Treat program, which streamlined the process of getting a prescription from a doctor by making it possible for people to test themselves and then go to their local pharmacy where the pharmacist could write a prescription for COVID-19 antiviral medications if they were positive The availability of more self-tests means moving that entire process into the home, which would make testing and treating respiratory diseases more convenient and streamlined, which in turn could reduce their spread.
More From TIME
eMed is working with the National Institutes of Health to study what impact having more at-home tests will have in controlling spread of disease. The team is tracking the average time it takes for people who test positive using eMed’s at-home kits—combined with a telehealth visit on eMed’s platform—to get treatments compared to people who make appointments with their doctor or visit a pharmacy. NIH is funding the test kits and telehealth visits for 80,000 to 100,000 people who enroll in the study through June 2024.
Mina argues that wider testing, driven by at-home kits and telehealth, could lead more people to reach out to their health care professionals for the proper treatment, which in turn could keep spread of infectious diseases down, and lower hospital costs for those who develop complications. “A $25 home testing kit and telemedicine visit could avoid what could turn into an $8,000 hospital visit,” says Mina.
The negatives of at-home testing
There is a downside to more DIY testing, however. The results are essentially invisible to public health authorities, so they don’t have a clear picture of how many people are getting sick, and where. If at-home testing, especially for infectious diseases, continues to grow, then public health officials will have to look beyond lab-based results for alternative ways to track the ebb and flow of cases, such as relying on anonymous sampling of wastewater from communities.
The future of at-home testing
Researchers are continuing to work on improving the accuracy, convenience, and price of at-home tools to test for infections. They are developing cheaper versions of rapid antigen tests that utilize paper strips (similar those used for urinary tract infection tests) that could bring the price of these tests down from over $20 to a few dollars. These, and other rapid antigen tests, will continue to be useful for people who don’t have symptoms but want quick answers to whether they can safely visit loved ones in the hospital without putting them at risk—or, if they have symptoms, whether they should go to work or school if they aren’t feeling well. “If your kids are sick and you want to test them before sending them to school and causing a flu outbreak, you don’t need perfect tests when you’re trying to be a good citizen,” says Mina. “Rapid antigen tests that are 80% or more sensitive are amazingly important for public health.”
Read more: What to Know About the New COVID-19 Vaccine Booster
PCR-quality tests are also becoming cheaper and more effective. The FDA recently issued an emergency use authorization for a test made by Aptitude, called Metrix, that mimics PCR by relying on electrochemical signals from saliva or nasal swab samples that can pick up viral genes in about 30 minutes. The reader device, which can be reused, costs $75 and a single-use saliva test kit for COVID-19 is $48. Researchers are working on cheaper versions of these molecular tests, and some may eventually be read through devices that attach to smartphones.
“The biggest challenge continues to be price,” says Dr. Carlos del Rio, president of the board of directors of the Infectious Diseases Society of America and professor of medicine at Emory University. “We need to hopefully see the price come down like they have for some cell phones and computers.” Even the lowest-priced option, the rapid antigen tests, are currently in the range of $25 per test—which can add up, especially when families need to buy several kits to test everyone multiple times in a household.
Eventually, even PCR-based devices may become small, convenient, and cheap enough for people to have at home. Such diagnostic testing may follow the example of pregnancy testing, which for decades could only be done at a doctor’s office with blood and urine samples and can now be done at home with an inexpensive urine stick. “I truly believe that within five years we will have little molecular devices in our homes that can test for a panel of five things,” says Dr. Luis Ostrosky, chief of infectious diseases at UTHealth Houston and Memorial Hermann. “We’re living in a revolution in diagnostics.”
More Must-Reads from TIME
- How Donald Trump Won
- The Best Inventions of 2024
- Why Sleep Is the Key to Living Longer
- Robert Zemeckis Just Wants to Move You
- How to Break 8 Toxic Communication Habits
- Nicola Coughlan Bet on Herself—And Won
- Why Vinegar Is So Good for You
- Meet TIME's Newest Class of Next Generation Leaders
Contact us at letters@time.com