By David Stout
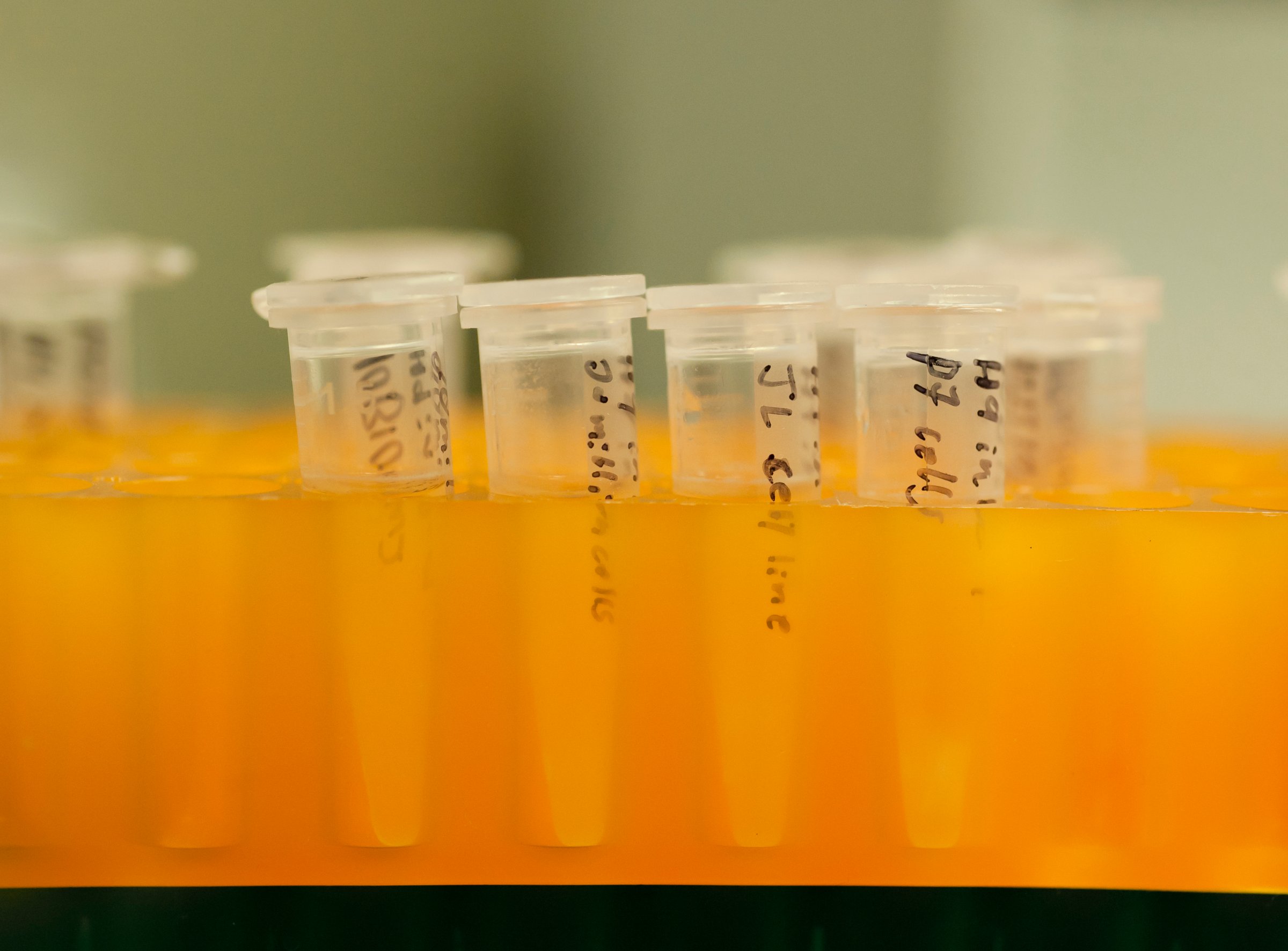
The U.S. Food and Drug Administration has allowed Google-backed company 23andMe to begin marketing a home genetic test for Bloom Syndrome — an inherited condition characterized by shortness of height and increased risk of cancer.
The FDA also announced that it intends to exempt similar genetic testing devices from its premarket review protocol.
Screening tests are largely used by prospective parents who are concerned that their future children may inherent harmful genetic disorders.
“Today’s authorization … along with FDA’s intent to exempt these devices from FDA premarket review, supports innovation and will ultimately benefit consumers” stated spokesman Alberto Gutierrez.
More Must-Reads from TIME
- How Donald Trump Won
- The Best Inventions of 2024
- Why Sleep Is the Key to Living Longer
- Robert Zemeckis Just Wants to Move You
- How to Break 8 Toxic Communication Habits
- Nicola Coughlan Bet on Herself—And Won
- Why Vinegar Is So Good for You
- Meet TIME's Newest Class of Next Generation Leaders
Contact us at letters@time.com