A drug normally used to treat bone marrow disorders may help patients suffering from alopecia, according to a new study.
Alopecia areata is a kind of autoimmune disorder that causes the immune system T-cells to attack hair follicles, causing them to fall out and become dormant. For some sufferers, this means the loss of small patches of hair, such as “spot baldness”, but a small percentage suffer complete hair loss. No current treatments for alopecia patients completely restore hair loss.
For a long time, medical researchers were uncertain what was causing the immune system cells to attack hair follicles, until Columbia University Medical Center (CUMC) researchers were able to determine a “danger signal” in the follicles of alopecia patients that was signaling the T-cells’ attack.
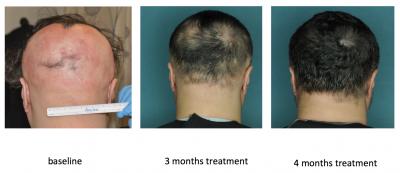
Now, they’re moving on to search for a cure. In the paper, published in Nature Medicine, the researchers report their early results in both mice and human trials. The researchers tested two FDA-approved JAK inhibitors (or involved in immune response), ruxolitinib and tofacitinib. In mice with severe hair loss they found that both of the drugs completely restored the hair in the animals. In three human patients in the researchers’ clinical trial of ruxolitinib, hair loss was restored within four to five months. Ruxolitinib was already approved by the FDA as a treatment for myelofibrosis.
One of the lead doctors on the clinical trial is Angela M. Christiano, a professor in the Departments of Dermatology and of Genetics and Development at CUMC, who is herself an alopecia sufferer. “Patients with alopecia areata are suffering profoundly, and these findings mark a significant step forward for them,” she said in a statement. “The team is fully committed to advancing new therapies for patients with a vast unmet need.”
The findings are early, but given the limited amount of research and resources dedicated to the study of alopecia, the findings will be a great relief to sufferers—especially as the drug in question has already been given the thumbs up by the FDA.
More Must-Reads From TIME
- The 100 Most Influential People of 2024
- Coco Gauff Is Playing for Herself Now
- Scenes From Pro-Palestinian Encampments Across U.S. Universities
- 6 Compliments That Land Every Time
- If You're Dating Right Now , You're Brave: Column
- The AI That Could Heal a Divided Internet
- Fallout Is a Brilliant Model for the Future of Video Game Adaptations
- Want Weekly Recs on What to Watch, Read, and More? Sign Up for Worth Your Time
Contact us at letters@time.com