I have two children. At the moment, all I know about their genes is that they both have 46 chromosomes, and one is XY and one is XX.
I try to treat them equally, to assume equal potential. But what if I knew my daughter carried a “smart” gene and my son did not? When he came home from school with a B, would I assume it was just his genes, and not push him to try harder? And what if I could have known this before he were born, at a time when he was just a little blip on an ultrasound? Frankly, I’m not sure I would trust myself with this information.
Such knowledge isn’t, of course, possible yet. For one thing, we haven’t yet found many genes that can reliably predict intelligence. And at the moment, even if we did know what genes we were looking for, we wouldn’t be able to find them very early in pregnancy. But thanks to a new kind of fetal genetic testing this may be starting to change.
Once upon a time, everything about your baby was a surprise until the moment of birth. Is it a boy or a girl? Does he (or she!) have all 10 fingers and 10 toes? And, most important: is the baby healthy? Genetic disorders—Down Syndrome, Trisomy 18 and others—were often a surprise in the delivery room.
We may still engage in the ritual counting of fingers and toes on our new baby, but it’s all for show: really, we checked for those months ago in an ultrasound. And for many women, genetic testing during pregnancy has ruled out—or all but ruled out—the possibility that their child has a genetic abnormality.
Recently, the introduction of “cell-free fetal DNA testing” has altered the landscape of prenatal genetic testing further. You may have heard of these tests by their brand names: Harmony or MaterniT21, among others. The technology for each is broadly the same: they rely on a sample of maternal blood with no risk to the fetus, and they have accuracy rates approaching those of fetal diagnostic test like amniocentesis or Chorionic Villus Sampling (CVS), but without any risk to the fetus. In other words, these new tests provide the best of both worlds—and a recipe for moral fission.
The Evolution of Fetal Screening
Prenatal genetic testing, imperfect though it has been, is not new. Beginning in the 1970s, amniocentesis allowed doctors to identify genetic disorders in utero, typically mid-pregnancy. Within the following decade, CVS provided an alternative to amniocentesis that could be performed earlier in pregnancy—in the first trimester rather than the second—and provide similar information. These two procedures provide complete genetic information on the fetus—labs can literally sequence its entire genome.
This means that although these are most commonly used to detect the most common genetic disorders—Down Syndrome, for example—they could, in principle, be used to detect more minor genetic abnormalities, or even to identify normal genomic variations, such as a predisposition for having red hair. (Such uses are rare, mind you, since both procedures are invasive and carry some small risk to the fetus.) Historically, the alternative has been prenatal screening that relied primarily on an ultrasound, which isn’t risky to the fetus but also cannot concretely diagnose a problem, thus, they must be followed up by one of the invasive procedures if a problem is suspected.
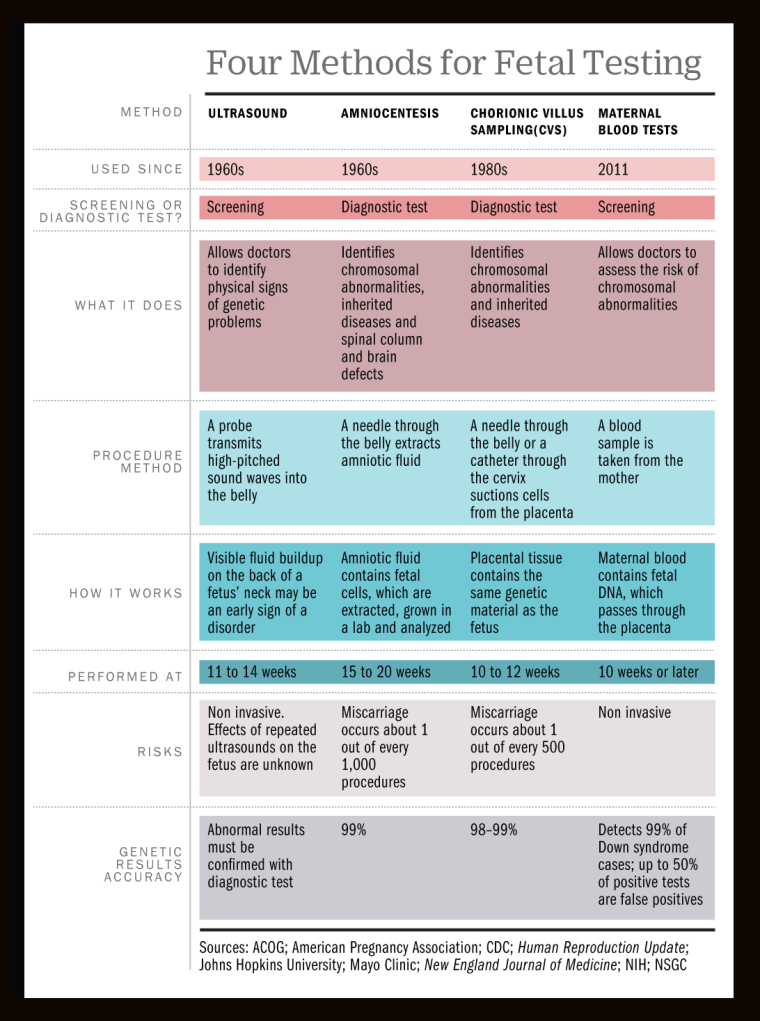
The fact that we can determine characteristics of a baby in utero, combined with the availability of abortion, has always made some people nervous. It suggests eugenics, or a future of “designer babies.” This is not all fanciful concern: in some countries prenatal sex determination and sex-selective abortion have altered the overall sex ratios in the population (for example, in recent years in China, 120 boys are born for every 100 girls). But the fact is that, in general, and in the U.S. in particular, prenatal screening has been effectively limited to serious genetic disorders—the risk of harming the fetus has outweighed the value of the information for other uses.
But the new prenatal screening tests are a game changer. They represent a significant technological breakthrough because the key to identifying problems or genetic risks is being able to see the baby’s DNA. Amniocentesis and CVS accomplish this with amniotic fluid or placental material, both of which contain fetal cells and, hence, fetal DNA. But that means going inside the womb—with a needle, typically—and actually taking some cells. And that’s what carries the risk.
Some fetal cells also circulate in the maternal bloodstream during pregnancy—that’s not news—it’s just that the volume of fetal cells in maternal blood is very low, making them difficult to use in a practical way. The technological breakthrough was the recognition of “cell-free fetal DNA”—that is, fetal DNA outside of cells.
When cell-free DNA is isolated in maternal plasma, 10% to 20% of it is fetal in origin. In lay terms, this means that researchers can be confident that a large share of what they are extracting comes from the fetus. In principle, if one could simply separate the maternal and fetal DNA, it would be possible to sequence the full fetal DNA using this procedure.
Technology is still not quite there yet, but this procedure currently works by looking for things in the cell-free DNA which wouldn’t be there if the DNA it were just the mother’s.
Think about it in terms of gender: Women have two X chromosomes; men have one X and one Y. Imagine you look in mom’s cell-free DNA and you find a bunch of Y chromosomes. The baby will be a boy, right? If you don’t see any Y chromosomes, a girl.
Similarly, a fetus with Down Syndrome has three copies of chromosome 21, rather than two, but two copies of all of the other chromosomes. So if you look at a mix of fetal and maternal DNA together, from a genetically normal mother, and see relatively more copies of chromosome 21, you would suspect the baby has Down Syndrome. If any chromosomal imbalance is striking enough, the test results will flag a potential problem.
At the moment, these tests fall short of what is possible with amniocentesis or CVS testing. One way in which they fall short is they focus on only the three most common trisomies: Down syndrome (trisomy 21), Trisomy 18 and Trisomy 13. Invasive testing will detect other trisomies, and can detect other types of chromosomal problems as well. Another shortcoming: both false negatives and false positives are possible.
This procedure relies on a statistical threshold test: Sufficiently imbalanced, and the test pings “positive.” Not sufficiently imbalanced, it comes up negative. However, sometimes the imbalance in the chromosome counts isn’t striking enough to flag as a positive test, even when the baby does have a chromosomal abnormality. This is what is called a “false negative.” And on the other side, sometimes the chromosomes look imbalanced in the sample but the baby is fine. This is what is called a “false positive.”
False negatives are pretty rare in these new blood tests—for a woman in her early 30s with a negative result on this screening, the chance of a baby with a chromosomal abnormality is about 1 in 90,000. False positives on this test are also limited, but they matter more. For that same woman in her early 30s with a positive test result, the chance of having a baby with a chromosomal problem is about 66%. In other words, one out of three women who receive results indicating abnormalities will actually have a baby who is genetically normal.
Answering—and Raising—More Questions
Experts agree that consequential decisions about a pregnancy should not be made without an invasive test as a follow-up. But this is likely to be a temporary issue. Effectively, the problem is one of genetic sequencing capacity and statistics. Already these tests are close to perfect on detection of gender. And the precision with which genetic predictions can be made will also improve. It seems unlikely we are more than a few years away from the ability to use these tests as diagnostic.
As these tests improve, so too will the range of conditions they can detect. Researchers last year reported on a case in which they used a version of this test to detect a small genetic issue called a microdeletion. The impact of this microdeletion, which was passed on from the mother, is an increased risk for nearsightedness and mild hearing loss. The mother in the study learned that she was passing on her poor eyesight and bad hearing to her child.
In principle, this technology could be used to detect anything for which we have a known genetic link. Researchers engaged in Gene-Wide Association Studies (GWAS) have, in the past few years, made progress on identifying a few genes which code for intelligence. Imagine you’ve tested yourself and you know you carry one of these intelligence genes but, sadly, your spouse does not. Now imagine you can easily learn if your fetus got your smart genes, or your spouse’s not-so-smart ones. Or your genes for height, your risk for obesity, and your spouse’s gene for stubbornness (okay, we haven’t found this one yet).
Now take it a step further. Fetal DNA begins to circulate in the mother’s blood at the very start of pregnancy. At the moment, these tests wait until 10 or 11 weeks of pregnancy so the concentration of fetal DNA is high enough to use for accurate detection. But as the sequencing and statistics improve, we may find that it is possible to do the same testing at 8 weeks. Or six.
What if you could know, at six weeks of pregnancy, whether your child would inherit your height, or hair color, or IQ? As I mentioned earlier, early gender testing is already used for gender-selective abortion, largely outside the U.S. This was true even when gender detection was not possible until 18 or 20 weeks.
These technologies will raise questions far beyond gender. Many people terminate a pregnancy when they learn the fetus has Down Syndrome. What about learning that the child will have autism? Or simply that their IQ is likely to be below average? We are holding Pandora’s box. Once we open it and let the information out, we lose control over what it is used for.
I would argue there are further implications. Let’s say I find out my fetus has an increased genetic risk for obesity, and I ultimately have that child. How will I treat her? Will I obsess about everything she eats, every ounce of baby fat that doesn’t immediately melt away? Will she grow up to be obese, or have an eating disorder I was party to with my worry? Could this actually make things worse rather than better? The idea that more information is better relies on our ability to ignore it. But is this something that, as a parent, you could ever really ignore?
I’m trained as an economist, and one of our general principles is that more information is better. Information helps us make better—more optimal—decisions. And, crucially, more information cannot make you worse off, since you can always just ignore it. Under this theory, these advances in genetic testing should be welcomed without reservation.
In many dimensions, the improvements in testing bring only good. The ability to more accurately detect serious genetic conditions earlier in pregnancy allows women and their partners to make difficult decisions about pregnancy termination earlier in the pregnancy when the medical complications are less significant.

The balance between the values of information and the possibility of mis-use is a difficult one. It would be a shame to fail to pursue technologies that are likely to deliver great gains. At the same time, it is naïve to pursue them without thinking about their consequences. And we should start thinking about these now.
Ready or not, the future is coming.
Emily Oster is a professor of economics at Brown University and the author of Expecting Better
