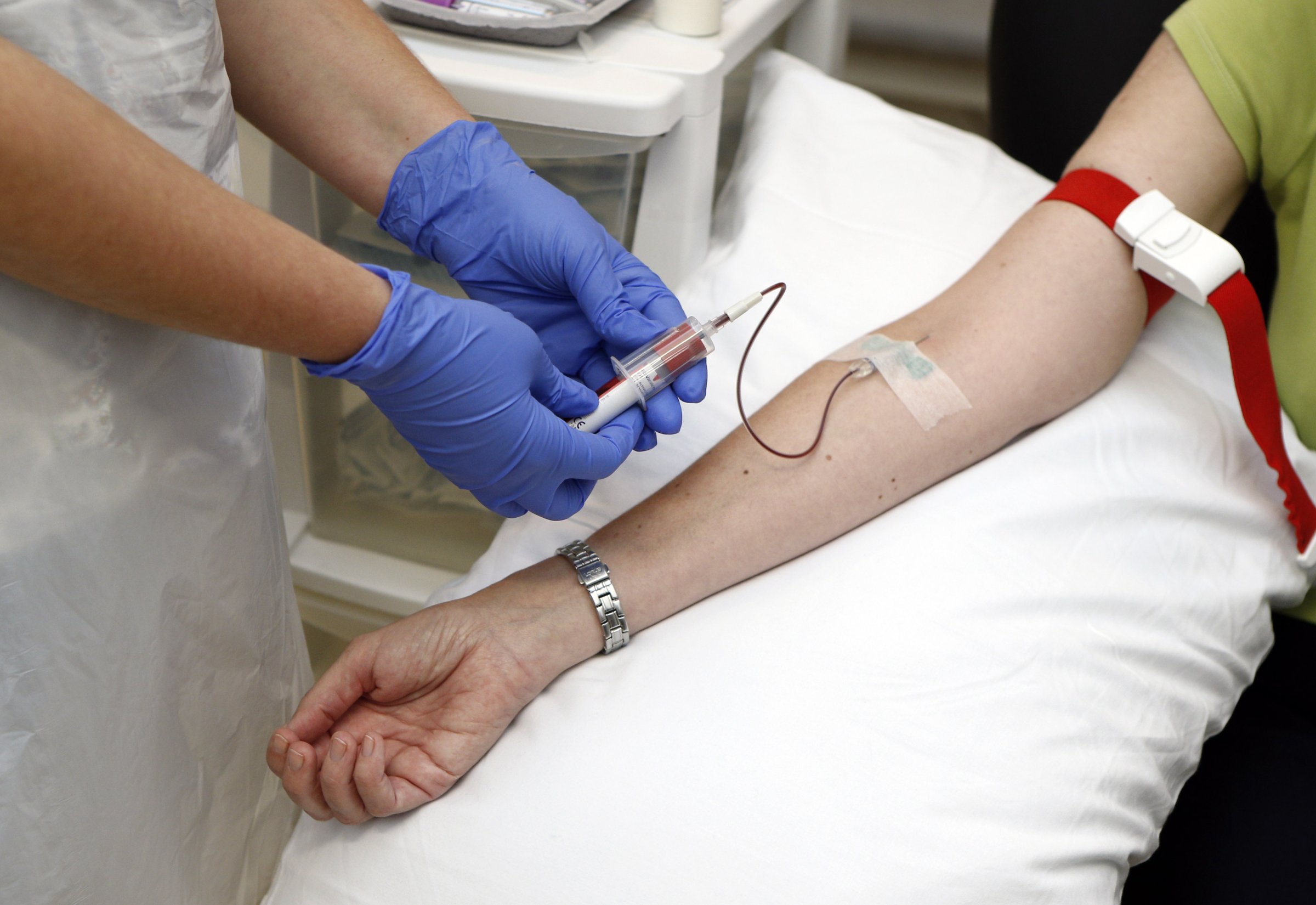
The world’s leading drugmakers are collaborating to develop millions of doses of an Ebola vaccine for use next year, aiming to prevent West Africans and frontline healthcare workers from contracting the deadly virus.
Johnson & Johnson said Wednesday that its goal is to produce 1 million doses of a two-step vaccine next year, and is already collaborating with Britain’s GlaxoSmithKline, Reuters reports.
The two companies could combine their vaccines and support each other’s work, while other companies are volunteering to provide production capacity. “I have spoken with (GlaxoSmithKline chief executive) Andrew Witty over the past few days several times as colleagues on how we are going to solve this,” Johnson & Johnson U.S. research chief Stoffels told reporters. “It might even be that we have to combine their vaccine with ours.”
The World Health Organization wants tens of thousands of people in West Africa, including frontline healthcare workers, to start receiving Ebola vaccines in January as part of clinical trials.
[Reuters]
More Must-Reads From TIME
- The 100 Most Influential People of 2024
- Coco Gauff Is Playing for Herself Now
- Scenes From Pro-Palestinian Encampments Across U.S. Universities
- 6 Compliments That Land Every Time
- If You're Dating Right Now , You're Brave: Column
- The AI That Could Heal a Divided Internet
- Fallout Is a Brilliant Model for the Future of Video Game Adaptations
- Want Weekly Recs on What to Watch, Read, and More? Sign Up for Worth Your Time
Contact us at letters@time.com