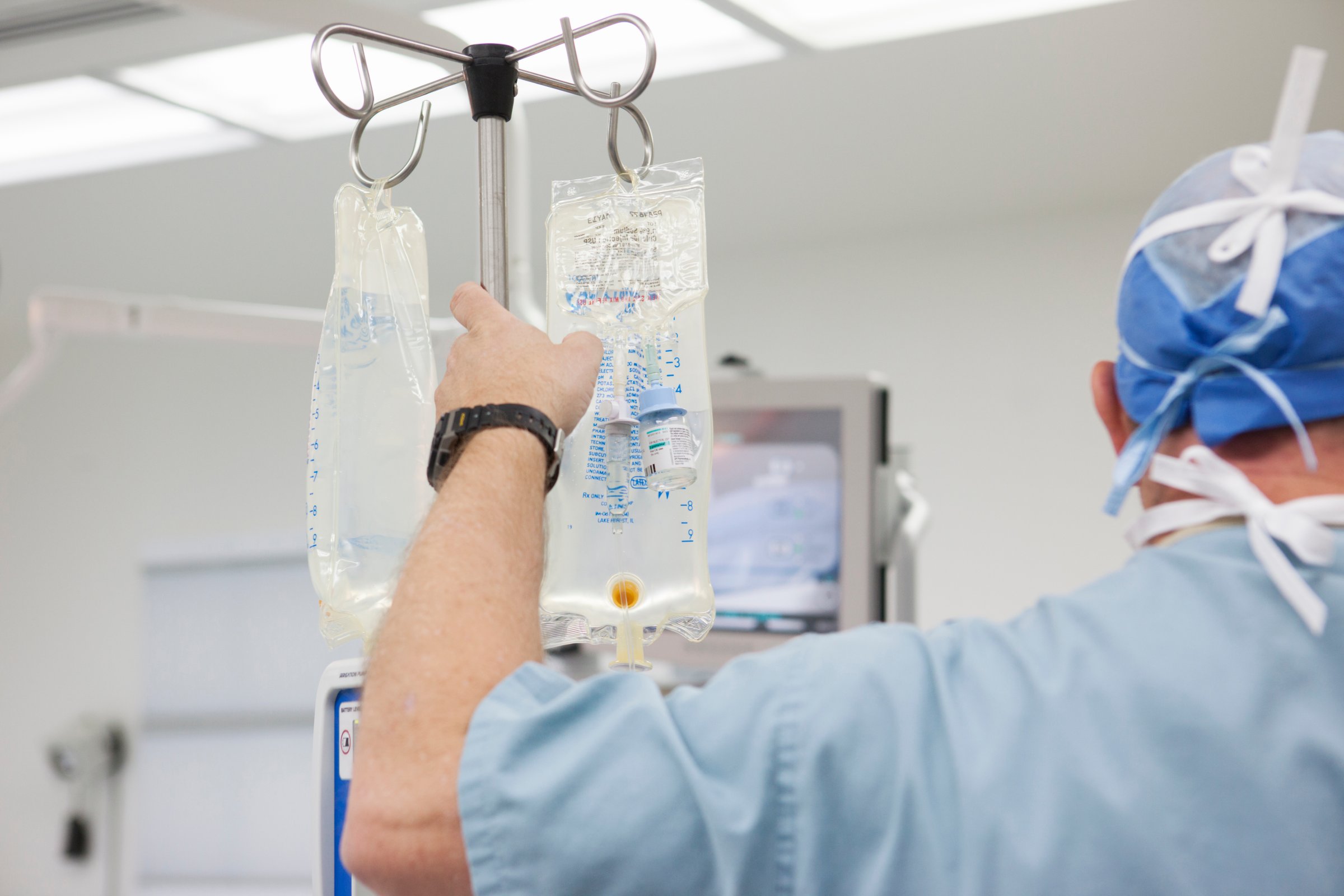
The U.S. Food and Drug Administration (FDA) warned hospitals and health care workers of a national shortage of intravenous (IV) saline solution, and urged doctors to conserve existing supplies. The agency is working with three manufacturers to address problem and provide alternatives if needed.
Millions of bags of the solution, which are essential for hydrating patients, and are also critical for cleansing the blood of dialysis patients, are used nationwide each week. The Washington Post reports that manufacturers told the FDA last year that they were expecting delays in filling hospitals’ orders after some plant shutdowns for routine inspection and upgrades. The IV solution cannot be replaced with other forms of saline because it is specifically designed to be administered by injection; in a letter to health care providers, the American Society of Health-System Pharmacists warned that saline solutions used for irrigation are “not approved for injection because it may have higher levels of particulates.”
MORE: Inside America’s Drug Shortage
The FDA says the shortage was triggered by several factors, including delays in delivery and increased demand by hospitals to treat flu patients.
Some healthcare providers are asking staff to find ways to conserve the IV solutions by using smaller bags, while others are using alternatives such as oral hydration fluids. Valerie Jensen, an associate director of the drug shortages program at the FDA told The Washington Post that no hospitals have actually run out of saline, they are “still reporting that they may only have a few days supply.” The agency is hoping the three major manufacturers of the product — Baxter Healthcare Corp., B.Braun Medical Inc., and Hospira Inc. — will be able to meet the increased demand in coming weeks.
More Must-Reads From TIME
- The 100 Most Influential People of 2024
- Coco Gauff Is Playing for Herself Now
- Scenes From Pro-Palestinian Encampments Across U.S. Universities
- 6 Compliments That Land Every Time
- If You're Dating Right Now , You're Brave: Column
- The AI That Could Heal a Divided Internet
- Fallout Is a Brilliant Model for the Future of Video Game Adaptations
- Want Weekly Recs on What to Watch, Read, and More? Sign Up for Worth Your Time
Contact us at letters@time.com